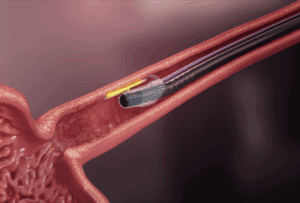
MiVu™ Mucosal Integrity Testing System
MiVu provides a complete picture of mucosal integrity with real-time results during a routine endoscopy. This revolutionary technology instantly differentiates between GERD, EoE, and Non-GERD, empowering physicians to improve clinical efficiency and enhance patient care.
100% Sensitivity for EoEvi
96% Positive Predictive Value for Refluxviii
95% Specificity for GERDviii
What is Mucosal Integrity Testing?
Mucosal integrity is influenced by dilated intercellular spaces (DIS), a key histologic biomarker of GERD and EoE that inversely correlates with mucosal integrity measurements. MiVu measures electrical impedance along 12 cm of the esophageal lining, providing a real-time display of mucosal integrity contour pattern and disease probability.
MiVu assesses esophageal mucosal integrity, which directly correlates with tissue damage caused by reflux and eosinophilic esophagitis. Unlike pH testing, which captures only acid reflux events that reach a specific level of the esophagus at a single point in time, MiVu detects long-term changes in esophageal health.
This cutting-edge approach may minimize false negatives and reduce the diagnostic uncertainty often seen with traditional pH testing.
Watch MiVu in Action During Endoscopy
See how MiVu integrates seamlessly into a routine endoscopy with the Endo Cap attached to the distal end of the endoscope.
When the Endo Cap is properly positioned against the mucosal lining, it captures immediate readings across 12 cm of the esophagus.
The Components of MiVu Mucosal Integrity Testing System

PriZm® Cart System
The system consists of the following components:
- PriZm Central Unit
- 24” LCD medical grade touch screen
- Keyboard & Mouse
- Computer
- Printer
- Ergonomic cart
- Zvu® Functional GI Software*
*Zvu is utilized for esophageal manometry, mucosal integrity testing, and reflux monitoring.

MiVu Esophageal Endo Cap
It is placed on the endoscope tip and is required to perform MiVu testing.
- A single-use Endo Cap is compatible with an endoscope diameter of 9.9 mm.

MiVu Cable
Connects the Endo Cap to the PriZm Central Unit
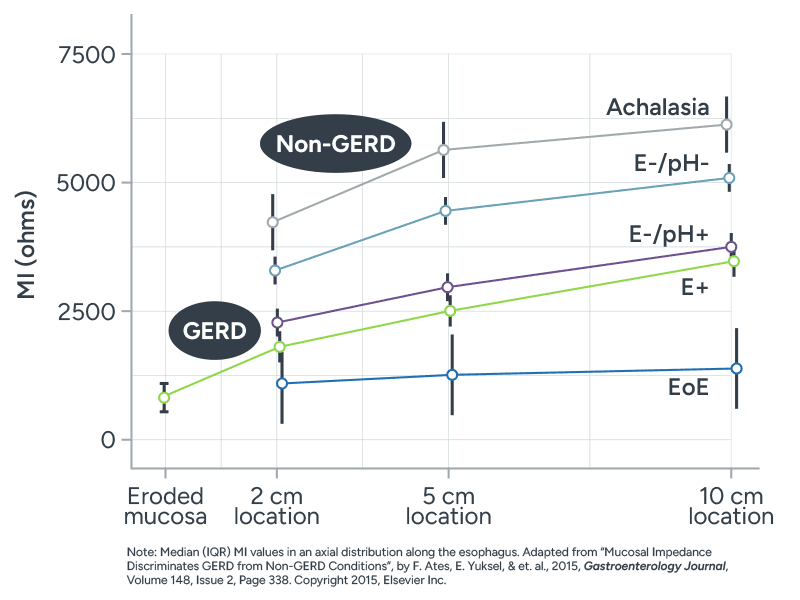
MiVu Simplifies Assessing Esophageal Diseases
This chartviii illustrates how mucosal impedance values differ for various esophageal conditions.
- Non-GERD patients typically tend to have higher impedance (ohms) values across the entire esophagus, indicating a normal mucosa.
- Patients with active EoE consistently show lower impedance across all measured points, reflecting mucosal damage along the entire esophagus.
- Esophagitis patients tend to have lower impedance values closer to the gastroesophageal junction (GEJ), which gradually increase and normalize proximally. This same pattern exists for individuals without esophagitis, but positive pH.
Discover more about MiVu, including its clinical application in daily practice, with a recent on-demand webinar on our Diversatek University platform.
What are the Benefits of Using MiVu?
MiVu empowers gastroenterologists with real-time data to aid in the differentiation of GERD, EoE, and Non-GERD. By simplifying and enhancing diagnostic workflows, MiVu improves procedural efficiency and reduces diagnostic latency.
MiVu vs Traditional pH Monitoring?
MiVu has numerous benefits when compared to traditional ambulatory reflux monitoring, including:
- Aid in the detection of GERD and EoE in minutes
- May reduce false negatives and inconclusive results
- Reduce patient discomfort & eliminate the need for patient compliance
- Integrate seamlessly into your current endoscopy workflow
| Feature | MiVu | Traditional pH Monitoring |
|---|---|---|
| Specificity for GERDviii | 95% | 64% |
| Positive Predictive Valueviii | 96% | 40% |
| Test Time | 2 Minutes | 24-48 Hours |
| Measures | Mucosal integrity & reflux impact | Only acidity (pH) |
| Detects Non-Acid Reflux? | Yes | No |
| Sensitivity for EoEvi | 100% | Not designed for EoE |
| Patient Friendly? | Single Procedure | Requires catheter placement |
Review the Science
Access a comprehensive library of clinical evidence and publications in support of MiVu
-
i Dent J. Gastro-oesophageal reflux disease. Digestion 1998;59: 433–445.
-
ii Patel DA, Vaezi MF. Utility of esophageal mucosal impedance as a diagnostic test for esophageal disease. Curr Opin Gastroenterol 2017;33:277–284.
-
iii Wenner J, Johansson J, Johnsson F, et al. Optimal thresholds and discriminatory power of 48-h wireless esophageal pH monitoring in the diagnosis of GERD. Am J Gastroenterol 2007;102:1862–1869.
-
iv Kessels SJM, Newton SS, Morona JK, et al. Safety and efficacy of wireless pH monitoring in patients suspected of gastroesophageal reflux disease: a systematic review. J Clin Gastroenterol 2017;51:777–788.
-
v Caroline Barrett & Yash Choksi & Michael F. Vaezi, Mucosal Impedance: a New Approach to Diagnosing Gastroesophageal Current Gastroenterology Reports (2018) 20:33 Reflux Disease and Eosinophilic Esophagitis.
-
vi Choksi Y, Lal P, Slaughter JC, et al. Esophageal mucosal impedance patterns discriminate patients with eosinophilic esophagitis from patients with GERD. Clin Gastroenterol Hepatol 2018;16:664–671 e1.
-
vii Dhyanesh A. Patel, Tina Higginbotham, James C. Slaughter, Muhammad Aslam, Elif Yuksel, David Katzka, C. Prakash Gyawali, Melina Mashi, John Pandolfino, and Michael F. Vaezi, Development and Validation of a Mucosal Impedance Contour Analysis System to Distinguish Esophageal Disorders, Gastroenterology 2019;156:1617–1626.
-
viii Ates, F., Yuksel, E. S., Higginbotham, T., Slaughter, J. C., Jetté, M. E., Lee, A. S., ... & Vaezi, M. F. (2015). Mucosal impedance discriminates gastroesophageal reflux disease from non-gastroesophageal reflux disease conditions. Gastroenterology, 148(2), 334–343
-
MiVu™ Mucosal Integrity Testing System is covered by one or more of the following patents: US 11,291,382, US 10,321,867 and US 9,814,408.
Zvu® Functional GI Software
Zvu is a powerful, user-friendly software platform designed for acquiring, managing, and analyzing GI motility data. With customizable options, Zvu allows providers to tailor diagnostic testing to their procedural needs and preferences, consistently delivering reliable and easy-to-read results.
How to Read MiVu Data
Zvu Software visualizes ohms values utilizing distinct color patterns that facilitate interpretation and aid physicians in diagnosing GERD, EoE, and Non-GERD. This innovative testing system assesses 10 cm or more of the mucosal lining to quantify esophageal health.
Hover over the bars and the image to see how the data is presented.
This data acquisition screen displays the four required measurements in clearly defined columns for easier analysis.
GERD appears red, with lower ohms values, particularly red distally.
EoE shows red throughout the esophagus from top to bottom.
Non-GERD appears blue/yellow, indicating higher ohms values, which are associated with non-GERD.
Diversatek University (DU) offers comprehensive education and training, online, on-site, and in-person, to ensure your team receives a personalized learning experience that supports your goals and enhances clinical confidence.

MiVu Mucosal Integrity Testing System Fact Sheet
| Part No. | Product | Description |
|---|---|---|
| PRIZMCART-II | PriZm® Base System |
For new users who want to perform High Resolution Manometry or MiVu ™ Mucosal Integrity Studies, without an existing Base System |
| PRIZMSYS-II | PriZm® Base System |
For new users who want to perform High Resolution Manometry or MiVu ™ Mucosal Integrity Studies, without an existing Base System − and include an isolation transformer. |
| PRIZMUPG-KIT | PriZm® Base System |
For existing inSIGHT Ultima® Base Cart System (ULTCART) or inSIGHT Ultima® Base System (ULTSYS) users who want to perform High Resolution Manometry and have access to MiVu™ functionality. |
| MI-ESO-CAP-3L | MiVu™ Esophageal Endo Cap | |
| MI-CAB-02 | MiVu™ Endo Cap Cable | Connects the MiVu™ Esopahgeal Endo Cap to the PriZm Central Unit |
| ZVU-3 | Zvu® Software | |
| ZVU-3-ZIP | Zvu® Software |
| Part No. | Product | Description |
|---|---|---|
| COMP-KIT | Desktop PC with Windows | |
| L89-5500 | Desktop Color Printer | |
| L89-5355 | Printer USB Cable | |
| SYS-MONITOR | Monitor |
Upgrade Your inSIGHT® Ultima System to Enable MiVu
Current Diversatek Healthcare customers using the inSIGHT® Ultima System will need to purchase the Upgrade Option to incorporate MiVu into their practice. Alternatively, they may choose to completely transition to the PriZm® Cart System.
To begin the upgrade process, please contact your Diversatek Healthcare Territory Managers or Customer Support at orders@diversatekhc.com.